Matigen® HCM
Collagen-based tissue Graft/ Prosthesis

Collagen Tissue Supplement
- Classified as a collagen tissue supplement, the product is derived from porcine dermis through a decellularization process that removes all cellular components and residues while preserving the structural integrity of the extracellular matrix.
- Its natural three-dimensional, white-sheet collagen structure facilitates skin regeneration by supporting cellular adhesion and proliferation on the graft material.
- The dermal matrix is processed to completely eliminate immunogenic factors, "with no reported cases of infection over the past five years."
Virus Inactivation Technology
- GENEWEL’s proprietary virus inactivation technology has been validated by national authorities through comprehensive biological safety testing, ensuring robust viral clearance and product stability.
- The material demonstrates reduced inflammatory response due to its rapid engraftment compared with conventional products.
- Furthermore, strict quality control of raw materials reinforces the product’s safety, sterility, and hygiene.
Cross-Linked vs. Non-Cross-Linked HCM
Matigen® HCM is a cross-linked material. It is a bio-adhesive hydrogel, used for tissue regeneration, that uses a muscle extracellular matrix (MEM) as a cross-linking agent.
Cross-Linked Matigen® HCM
- Greater stability: The cross-linking process creates a more stable, networked structure that is thicker and more resistant to enzymatic breakdown.
- Longer-lasting: Its effects last longer than non-cross-linked materials, which is crucial for regenerative applications.
- Provides structure: The material provides stable physical support for cell growth and tissue regeneration.
- เพิ่ม biochemical cues: It helps mediate the recruitment and maturation of muscle cells, making it effective for muscle regeneration.
Non-Cross-Linked HCM
- Has a shorter lifespan in the body.
- Degrades more rapidly.
- Has a natural and biocompatible composition.

Three-dimensional structural images showing how the material enhances fibroblast infiltration and new blood vessel formation, thereby promoting natural integration.”
Benefits
- Helps accelerate tissue regeneration because fibroblasts and new blood vessels can easily penetrate the material.
- Reduces the risk of inflammation or material rejection the material is naturally accepted by the body and integrates with existing tissue.
- Enhances the strength and durability of regenerated tissue collagen and blood vessel formation improve the quality of the restored tissue.
- Suitable for medical applications such as implants, bone/tissue substitutes, and the repair of large
Research
Patent No. 1014105330000 - ACELLULAR TREATMENT FORMED OF A MATERIAL DERIVED FROM BIOLOGICAL TISSUES, A TREATMENT METHOD USING THE SAME, AND A BIOLOGICAL MATERIAL ACQUIRED THEREBY
Summary: A biological material contains collagen and elastin. The thickness and the tensile strength of the biological material are 10 um to 3 mm and 0.01-20 MPa, respectively, for transplantation and procedure. The biological material has superior biocompatibility, cell affinity, and immunocompetence.

Collagenase Degradation Test
Matigen® HCM refers to a collagen-based biomaterial used in Hypertrophic Cardiomyopathy (HCM) research, and its results in a collagenase degradation test would typically show reduced mechanical properties and structural breakdown over time due to the enzymatic activity.
How the test works:
- Collagenase exposure: The Matigen® HCM biomaterial is incubated with a collagenase enzyme.
- Enzymatic action: Collagenase breaks down the collagen within the biomaterial's matrix.
- Monitoring changes: The degradation is assessed using techniques like environmental Scanning Electron Microscopy (SEM) and video imaging to observe changes in the material's structure and mechanical behavior.
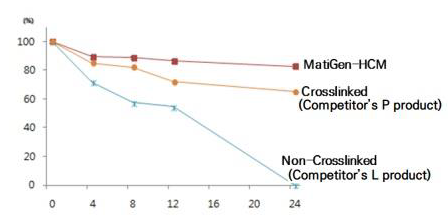
Comparing Matigen® HCM with two competitor products: a crosslinked ADM and a non-crosslinked ADM
Matigen® HCM (red line):
- Maintains the highest stability over time.
- After 24 hours, the material still retains close to 80–85% of its original structure.
- This indicates strong resistance to enzymatic breakdown.
Crosslinked Competitor’s Product (P) (orange line):
- Shows moderate stability.
- Degrades faster than Matigen® HCM but still retains around 60–70% at 24 hours.
- Crosslinking improves durability, but its performance is inferior to Matigen® HCM.
Non-Crosslinked Competitor’s Product (L) (blue line):
- Degrades rapidly under collagenase.
- Within 24 hours, most of the material is broken down (<10% remains).
- This suggests poor structural integrity and low enzymatic resistance.
Results
This testing provides crucial information for designing collagen-based scaffolds, like Matigen® HCM, that possess desired properties and appropriate degradation rates for use in tissue engineering.
Matigen® HCM demonstrates superior enzymatic stability compared to both crosslinked and non-crosslinked competitor products.
The data suggest that Matigen® HCM offers longer persistence in vivo, which is critical for effective tissue integration and regeneration.
The stability advantage likely translates to better clinical outcomes, including reduced resorption, more predictable graft survival, and improved patient safety.
